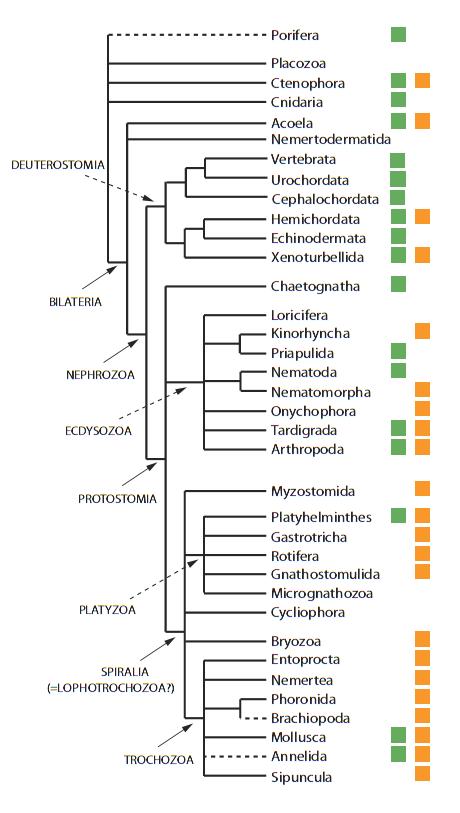
Within the Lophotrochozoa, no group of animals manifests these attributes as clearly as the Platyhelminthes: the diversity of body plans, developmental plasticity and ecological adaptations displayed by these flatworms is remarkable. This is despite the fact that it encompasses a collection of animals with an assortment of body plans, biological attributes, and ecological adaptations that is unmatched by the other two bilaterian metazoan branches combined. However, the third branch, the Lophotrochozoa, remains among the most undersampled and understudied collection of complex organisms on the planet. The second branch, the Ecdysozoa, is presently represented in biomedical research by the fruit fly Drosophila melanogaster and the roundworm nematode C. The Deuterostomes (the evolutionary lineage to which humans belong) are represented by a number of model organisms including mice, fish, sea squirts, sea urchins and, of course, humans. Now, in eLife, Christopher Laumer of Harvard University, Andreas Hejnol of the University of Bergen and Gonzalo Giribet, also from Harvard, have unravelled the phylogenetic tree of the Platyhelminthes, which are more commonly known as the flatworms ( Laumer et al., 2015).Īnimals possessing bilateral symmetry are presently grouped into three main branches in the metazoan tree of life. These considerations are particularly timely in view of current efforts to develop and characterize invertebrate model systems for studying regeneration and parasitism. Instead of selecting them purely on the biological attributes they conveniently exaggerate for our scientific interests, we could also consider the ecological and evolutionary histories that may have helped produce such attributes. As technology advances and allows us to examine aspects of biology that were not previously accessible to scientific interrogation, we may want to reconsider how we choose animals as new model systems. Our best chances of uncovering new biology and acquiring a truly transformative understanding of life are therefore to be found in the laboratory of nature.

Some have argued that further research into the model organisms that dominate much of biomedical research today could fill in the many gaps that exist in our understanding of life, but these organisms give a biased and ultimately poor statistical representation of the ∼30 million species of animals that populate our planet ( Brusca and Brusca, 2003). Paradoxically, while this approach has been remarkably successful in advancing our understanding of life, it has also made us acutely aware of how much more biology we have yet to comprehend.


For example, many model organisms produce large numbers of offspring, have features that make them easy to examine (such as transparent embryos), or are easy to domesticate and look after in the laboratory. Instead, organisms such as Drosophila, Caenorhabditis elegans, zebrafish and mice were primarily chosen as model organisms for purely practical reasons. The written accounts and reasoning associated with how the species we currently use for biomedical research were selected as model systems have rarely included weighty consideration of either their evolutionary or natural histories ( Alfred and Baldwin, 2015).
